In An Acidic Solution The
Consider the post-obit unbalanced redox reaction: Permit'south attempt to residuum this reaction in acidic solution. Fortunately, all atoms other than H and O are already counterbalanced, so we can move on to the next footstep. Because the number of electrons in the first half reaction (3e-) does not equal the number of electrons in the second one-half reaction (2e-), nosotros must multiply the reactions past coefficients so that the electrons volition cancel out when nosotros add together them. In this case, nosotros multiply the first reaction past 2 to yield 6e-, and the second reaction past 3 to yield 6e- as well. Carrying this out, we get: Now we can add the two reactions, and nosotros notice that the electrons on both sides cancel out. Thus, our counterbalanced reaction is: Now that we have learned how to residue redox reactions in acidic solution, we volition learn how to residue the aforementioned reaction in bones solution. Balancing redox reactions in basic solution is slightly more than complicated than balancing in acidic solution because nosotros must add both H+ and OH- to each half reaction. I will highlight the additional steps while we move through the procedure. Let'southward return to the same unbalanced reaction every bit above: We will try to residuum this reaction in basic solution. Fortunately, all atoms other than H and O are already balanced, then we can move on to the next stride. Nosotros simplify the equations to get: Recall that we must multiply the reactions by coefficients so that the electrons will cancel out when we add them. In this case, we multiply the starting time reaction past 2 to yield 6e-, and the 2nd reaction past 3 to yield 6e- too. Carrying this out, we become: Now we can add the two reactions, and we observe that the electrons on both sides cancel out. Thus, our last balanced reaction is: And there we have it! I promise you enjoyed this guide and found it helpful. For more practice on balancing redox reactions or other full general chemical science concepts, I recommend checking out Khan Academy, the UWorld MCAT Question Banking concern, or the ExamKrackers 1001 Questions in MCAT Chemistry workbook. If you are interested in working with me to set up for the MCAT, please attain out via the contact button beneath!-2.png?width=1080&name=Statistical%20Mediation%20%26%20Moderation%20in%20Psychological%20Research%20(1)-2.png) Balancing redox reactions is an essential skill for the Chemic and Physical Foundations department of the MCAT, the GRE Chemistry Subject Test, and the AP Chemistry Exam. Today, we will learn how to utilize the half-cell method for balancing redox reactions in acidic and basic solutions. We will first remainder a redox reaction in acidic solution, then we will balance the same redox reaction in basic solution.
Balancing redox reactions is an essential skill for the Chemic and Physical Foundations department of the MCAT, the GRE Chemistry Subject Test, and the AP Chemistry Exam. Today, we will learn how to utilize the half-cell method for balancing redox reactions in acidic and basic solutions. We will first remainder a redox reaction in acidic solution, then we will balance the same redox reaction in basic solution.Balancing Redox Reactions in Acidic Solution
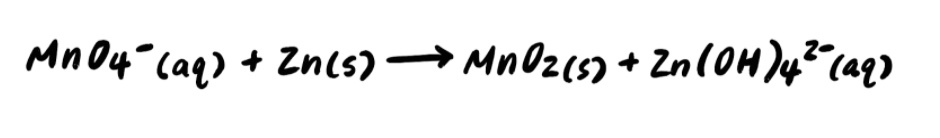
Pace 1: Split up the reaction into half reactions
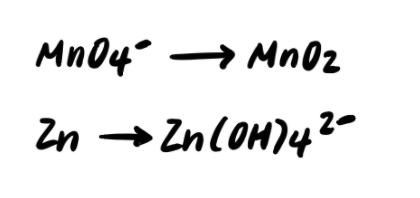
Step 2: Residue the elements other than H and O
Footstep three: Residual the O atoms by adding H2O
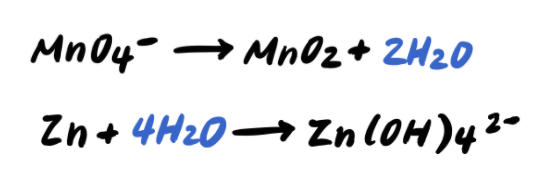
Step 4: Balance the H atoms by adding H+
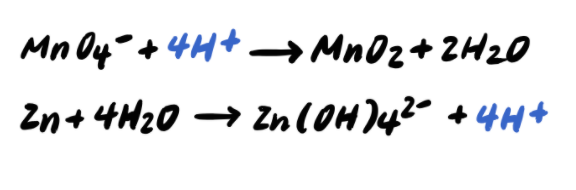
Stride 5: Residue the charges by adding eastward-
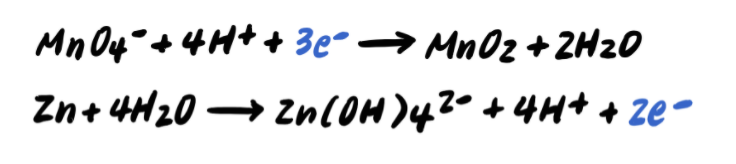
Step half dozen: Add the half reactions and simplify
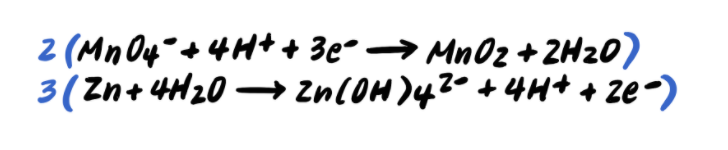

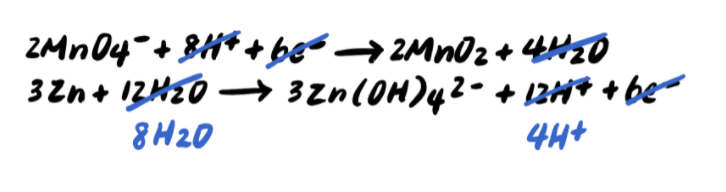
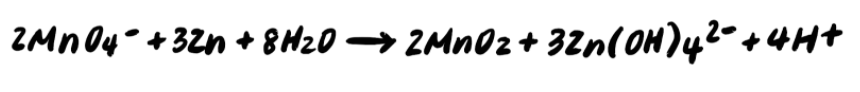
To summarize, the steps for balancing redox reactions in acidic solution are every bit follows:
Balancing Redox Reactions in Basic Solution

Step one: Split the reaction into half reactions
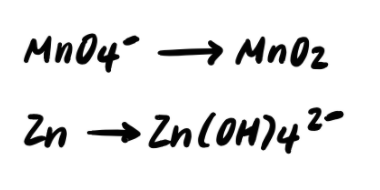
Stride 2: Balance the elements other than H and O
Footstep 3: Balance the O atoms by calculation Water
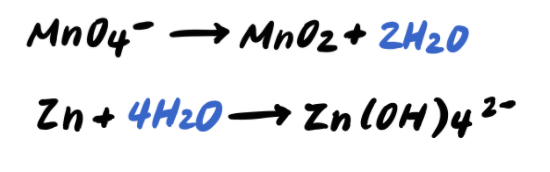
Step four: Balance the H atoms by adding H+
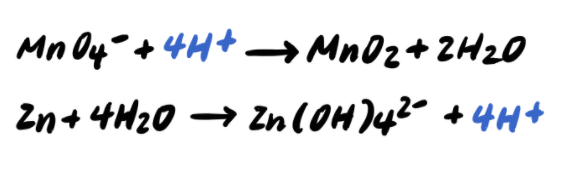
Pace five: Add OH- to BOTH SIDES neutralize any H+ [Boosted Step]
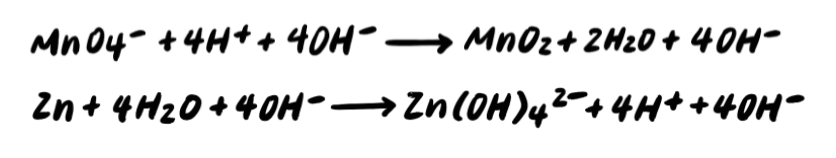
Pace 6: Combine H+ and OH- to brand H2O [Boosted STEP]
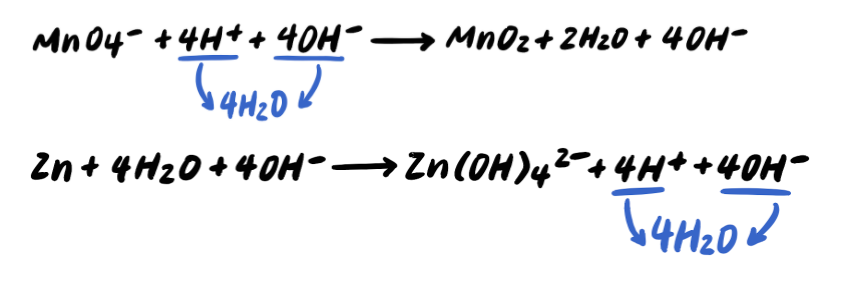
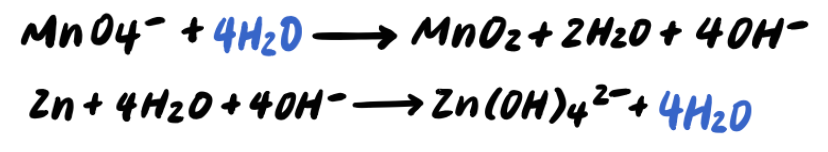
Step seven: Simplify by cancelling out excess H2o [ADDITIONAL STEP]
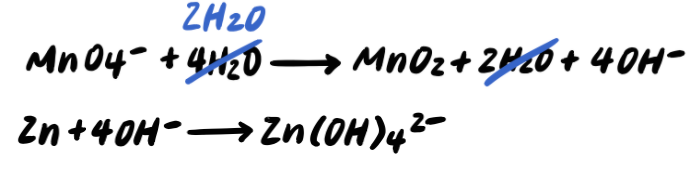
Stride eight: Rest the charges by adding due east-
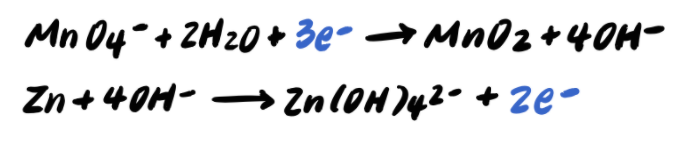
Pace 9: Add together the half reactions and simplify
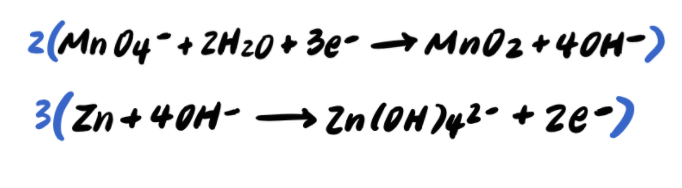
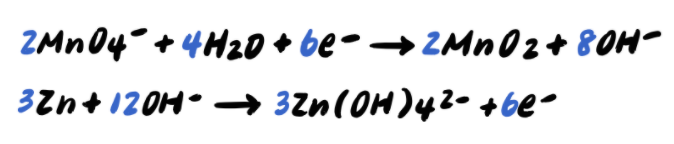
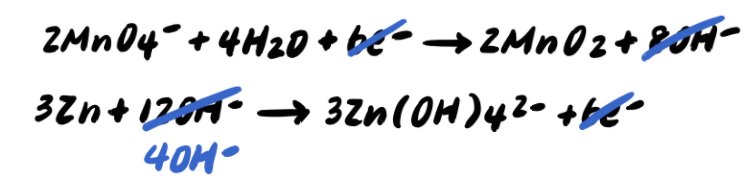
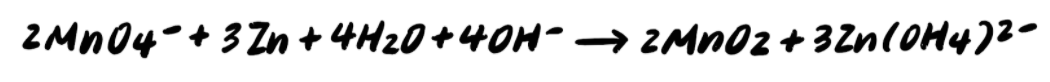
To summarize, the steps to balancing a redox reaction in basic solution are as follows:
In An Acidic Solution The,
Source: https://blog.cambridgecoaching.com/how-to-balance-redox-reactions-in-acidic-and-basic-solutions
Posted by: stringerbearring.blogspot.com


0 Response to "In An Acidic Solution The"
Post a Comment