How To Find Water Potential
17.1.i: Water Potential
- Folio ID
- 35218
Learning Objectives
- Define h2o potential and explain how it is influenced past solutes, pressure level, gravity, and the matric potential.
- Explicate which components of water potential plants tin manipulate and describe how.
Plants are phenomenal hydraulic engineers. Using simply the bones laws of physics and the simple manipulation of potential free energy, plants tin movement water to the pinnacle of a tree approaching 116 g (~381 ft, run into The Tallest Trees box). Plants tin also utilise hydraulics to generate enough force to separate rocks and buckle sidewalks (Figure \(\PageIndex{1}\)). Plants achieve this considering of h2o potential.

H2o potential is a measure of the potential energy in water. Plant physiologists are not interested in the energy in any one particular aqueous organisation, but they are very interested in water move between two systems. In practical terms, therefore, h2o potential is the divergence in potential energy betwixt a given water sample and pure water (at atmospheric force per unit area and ambient temperature). Water potential is denoted by the Greek alphabetic character ψ (psi) and is expressed in units of pressure (pressure is a form of free energy) chosen megapascals (MPa). The potential of pure water (Ψwestward pure H2O) is, by convenience of definition, designated a value of zippo (although pure water contains plenty of potential energy, that energy is ignored). Water potential values for the h2o in a establish root, stalk, or foliage are therefore expressed relative to Ψwestward pure H2O.
The h2o potential in plant solutions is influenced past solute concentration, force per unit area, gravity, and factors called matrix effects. Water potential tin be broken down into its individual components using the post-obit equation:
\[\psi_\text{arrangement} = \psi_\text{total} = \psi_s + \psi_p + \psi_g + \psi_m\]
where Ψs, Ψp, Ψone thousand, and Ψyard refer to the solute, pressure, gravity, and matric potentials, respectively. "System" can refer to the water potential of the soil water (Ψsoil), root h2o (Ψroot), stem h2o (Ψstem), leaf water (Ψleaf) or the water in the temper (Ψatmosphere): whichever aqueous system is nether consideration. As the private components change, they raise or lower the total water potential of a system. When this happens, water moves to equilibrate, moving from the system or compartment with a higher water potential to the system or compartment with a lower water potential. This brings the difference in water potential between the two systems (ΔΨ) back to cipher (ΔΨ = 0). Therefore, for water to move through the establish from the soil to the air (a process chosen transpiration), Ψsoil must be > Ψroot > Ψstem > Ψleaf > Ψtemper.
H2o only moves in response to ΔΨ, non in response to the individual components. Yet, because the individual components influence the full Ψsystem, by manipulating the individual components (especially Ψs), a plant can control water move. Solutes, pressure, gravity, and matric potential are all of import for the transport of h2o in plants. Water moves from an area of higher full water potential to an expanse of lower full water potential.
Solute Potential
Solute potential (Ψs), also called osmotic potential, is negative in a plant prison cell and nothing in distilled water. Typical values for cell cytoplasm are –0.5 to –1.0 MPa. Solutes reduce water potential (resulting in a negative Ψw) by consuming some of the potential energy available in the water. Solute molecules can deliquesce in h2o because water molecules tin can bind to them via hydrogen bonds; a hydrophobic molecule like oil, which cannot demark to water, cannot become into solution. The energy in the hydrogen bonds between solute molecules and water is no longer available to do piece of work in the system because it is tied upward in the bond. In other words, the amount of bachelor potential free energy is reduced when solutes are added to an aqueous arrangement. Thus, Ψs decreases with increasing solute concentration.
Because Ψs is one of the four components of Ψsystem or Ψtotal, a decrease in Ψs will cause a decrease in Ψtotal. Water moves towards areas of lower Ψsouthward (and thus lower Ψfull). In Effigy \(\PageIndex{2}\), the semipermeable membrane that separates the ii sides of the tube allows water but not solutes to pass. In the first tube, solute has been added to the right side. Calculation solute to the right side lowers Ψdue south, causing water to move to the correct side of the tube. Every bit a upshot, the water level is higher on the right side.
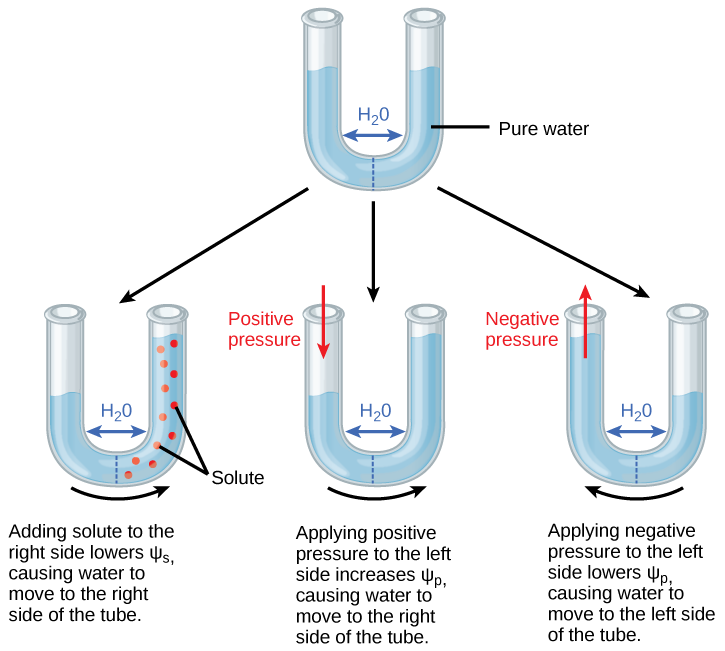
The internal water potential of a plant cell is more than negative than pure water considering of the cytoplasm'south loftier solute content. Because of this difference in water potential water will movement from the soil into a plant'due south root cells via the process of osmosis. This is why solute potential is sometimes called osmotic potential. Plant cells can metabolically dispense Ψs (and by extension, Ψtotal) past adding or removing solute molecules. Therefore, plants have control over Ψtotal via their ability to exert metabolic control over Ψsouthward.
Pressure Potential
Pressure potential (Ψp), also called turgor potential, may be positive or negative (Figure \(\PageIndex{c}\)). Because pressure is an expression of energy, the higher the pressure, the more potential free energy in a system, and vice versa. Therefore, a positive Ψp (pinch) increases Ψtotal, and a negative Ψp (tension) decreases Ψfull. The second tube in Figure \(\PageIndex{2}\)) has pure water on both sides of the membrane. Positive force per unit area is applied to the left side. Applying positive pressure level to the left side causes Ψp to increase. As a results, water moves to the right and then that the water level is higher on the correct than on the left. The third tube also has pure water, but this time negative pressure is practical to the left side. Applying negative pressure lowers Ψp, causing water to motility to the left side of the tube. Every bit a result, the water level is higher on the left.
Positive pressure inside cells is contained past the jail cell wall, producing turgor pressure. Pressure potentials are typically around 0.vi–0.8 MPa, but can accomplish as loftier as ane.5 MPa in a well-watered establish. A Ψp of 1.5 MPa equates to 210 pounds per square inch (1.5 MPa x 140 lb in-2 MPa-i = 210 lb/in-2). As a comparing, most car tires are kept at a force per unit area of 30–34 psi. An instance of the effect of turgor pressure level is the wilting of leaves and their restoration afterwards the plant has been watered (Figure \(\PageIndex{three}\)). Water is lost from the leaves via transpiration (budgeted Ψp = 0 MPa at the wilting indicate) and restored by uptake via the roots.

A found tin manipulate Ψp via its ability to manipulate Ψs and by the process of osmosis. If a found cell increases the cytoplasmic solute concentration, Ψdue south will decline, Ψtotal will decline, the ΔΨ betwixt the cell and the surrounding tissue will decline, water will move into the jail cell past osmosis, and Ψp volition increase. Ψp is likewise nether indirect plant control via the opening and closing of stomata. Stomatal openings allow water to evaporate from the leaf, reducing Ψp and Ψtotal of the foliage and increasing information technology between the water in the foliage and the petiole, thereby allowing water to catamenia from the petiole into the foliage.
Gravitational Potential
Gravitational potential (Ψg) is always negative to goose egg in a found with no pinnacle. The force of gravity pulls water downwards to the soil, reducing the departure in h2o potential betwixt the leaves at the peak of the plant and the roots. The taller the found, the taller the h2o column, and the more influential Ψg becomes. On a cellular scale and in short plants, this effect is negligible and easily ignored. However, over the height of a tall tree like a giant coastal redwood, the gravitational pull of –0.1 MPa m-1 is equivalent to an extra 1 MPa of resistance that must be overcome for water to attain the leaves of the tallest trees. Plants are unable to manipulate Ψg.
Matric Potential
Matric potential (Ψthousand) is always negative to zero. In a dry system, it tin can be as low as –2 MPa in a dry seed, and it is goose egg in a water-saturated system. The binding of h2o to a matrix always removes or consumes potential energy from the arrangement. Ψm is similar to solute potential because information technology involves tying up the energy in an aqueous system by forming hydrogen bonds betwixt the h2o and some other component. However, in solute potential, the other components are soluble, hydrophilic solute molecules, whereas in Ψm, the other components are insoluble, hydrophilic molecules of the plant cell wall. Every found cell has a cellulosic jail cell wall and the cellulose in the cell walls is hydrophilic, producing a matrix for adhesion of water: hence the name matric potential. Ψg is very large (negative) in dry tissues such equally seeds or drought-affected soils. However, it quickly goes to cypher as the seed takes up h2o or the soil hydrates. Ψgrand cannot be manipulated by the plant and is typically ignored in well-watered roots, stems, and leaves.
How To Find Water Potential,
Source: https://bio.libretexts.org/Bookshelves/Botany/Botany_(Ha_Morrow_and_Algiers)/Unit_3%3A_Plant_Physiology_and_Regulation/17%3A_Transport/17.01%3A_Water_Transport/17.1.01%3A_Water_Potential
Posted by: stringerbearring.blogspot.com



0 Response to "How To Find Water Potential"
Post a Comment